ABOUT THE PRINCIPAL INVESTIGATOR:
Dr. Shanmugaraju Sankarasekaran received his Ph.D.(Inorganic Chemistry) from the Department of Inorganic and Physical Chemistry, Indian Institute of Science, Bangalore and was awarded the Prof. S. Soundararajan Gold Medal for the Best Ph.D. Thesis in 2014. He received his IRC Postdoctoral Fellowship and Senior Postdoctoral Fellowship from the Trinity Biomedical Sciences Institute, Trinity College Dublin, Ireland and was honoured with the Best Poster Prize in Catalysis and Sensing for our Environment Symposium during his time there. Dr. Shanmugaraju was recently admitted as a member of the Royal Society of Chemistry(MRSC). He is now an Assistant Professor in Chemistry in IIT Palakkad with numerous publications under his name. Fluorescence and Visual Sensing of Nitroaromatic Explosives Using Electron-rich Discrete Fluorophores is one of them.
INTRODUCTION:
Volatile Organic Compounds (VOCs) are a large group of chemicals that are found in many products we use to build and maintain our homes. They are organic compounds that have high vapour pressures at room temperature. High vapour pressure correlates with a low boiling point, which in turn relates to a trait known as volatility. Volatility is a material quality which describes how readily a substance vaporizes. VOCs are used very frequently in manufacturing industries and scientific laboratories. Hence, they've become a major environmental pollutant. VOCs in general are not acutely toxic, but are known to have long-term chronic health effects. VOCs contribute to the formation of smog and also react with other air pollutants to produce ground level ozone which can damage crops and other materials. Since VOCs generally have a negative impact on the environment, it has become necessary to identify their presence in our surroundings.
One particularly notorious VOC is 1,4-dioxane. It has been known to pose a cancer risk to people. It dissolves in water and as a result is very capable of contaminating drinking-water facilities. Dioxane is also a trace contaminant in some cosmetic products such as perfumes, shampoos and toothpastes. It forms as a byproduct during the manufacturing process of certain cosmetic ingredients.
To alleviate environmental pollution, the search for a suitable chemosensor for the fast and selective sensing of hazardous substances and organic pollutants is attracting a lot of attention. Several sophisticated instrumental techniques are currently available for VOC detection and analysis, such as gas-chromatography and mass spectrometry. However, the usage of such techniques is limited due to reasons such as their expensive apparatus, lack of portability, time-consuming detection process, and complicated instrument standardization.
As of late, fluorescence quenching/enhancement-based sensing has become an effective and alternative detection method for VOCs due to its simplicity, high sensitivity, easy visualization and comparatively short response time. Fluorescence quenching refers to any process that decreases the fluorescence intensity of a sample, and fluorescence enhancement increases the fluorescence intensity of a sample. A lot of fluorescent sensors have been developed, however, it has been noted that background interference limits their practical use. Hence, there exists a need for developing a different method to detect VOCs.
Turn-on fluorescence sensing seems to be alluring because the sensing event can be easily detected, even by the naked eye sometimes. Turn-off fluorescence sensing isn't quite as fascinating, but the design for such sensing techniques isn't as challenging as it is for turn-on fluorescence sensing. Dr. Shanmugaraju and his team then considered the possibility of a solvatochromic fluorescent being capable of acting as an effective substitute for the sensing of VOCs.
So What'd the Team Do?
Using amino-1,8-naphthalimide Troger's base (TBNaps) as the foundation, the team set out on developing an efficient fluorescent sensor to detect and distinguish between VOCs that are structurally and chemically similar to each other. TBNaps are very interesting V-shaped chiral molecules that are strongly coloured and fluorescent due to charge transfer within the molecule. They display significant Stokes shift in different solvents, and thus they could be used for the sensing of structurally similar VOCs. Stokes shift is the difference in energy between positions of the absorption and emission spectra of the same electronic transition.
A new TBNap functionalized organic compound, TB-TZ-COP was synthesized. It was observed that this particular compound had the potential to act as a fluorescence sensor to discriminate between VOCs of similar structure. After studying quite a few of its physical and chemical characteristics, it was concluded that TB-TZ-COP is, in fact, a superior sensor. After confirming its capabilities, tests were conducted to observe if VOCs could be sensed as well. These tests were done by dispersing TB-TZ-COP in various common solvents and then measuring the fluorescence emission intensity of TB-TZ-COP in those solvents. A few solvents that were used in the testing include toluene, acetone, 1,4-dioxane, and methanol. TB-TZ-COP was found to be highly sensitive and reversible. Notably, the suspension of TB-TZ-COP in dioxane displayed a substantial enhancement in fluorescence intensity when compared to other organic solvents used in the experiments. Of all the VOCs used when testing, the largest emission enhancement was observed for dioxane.
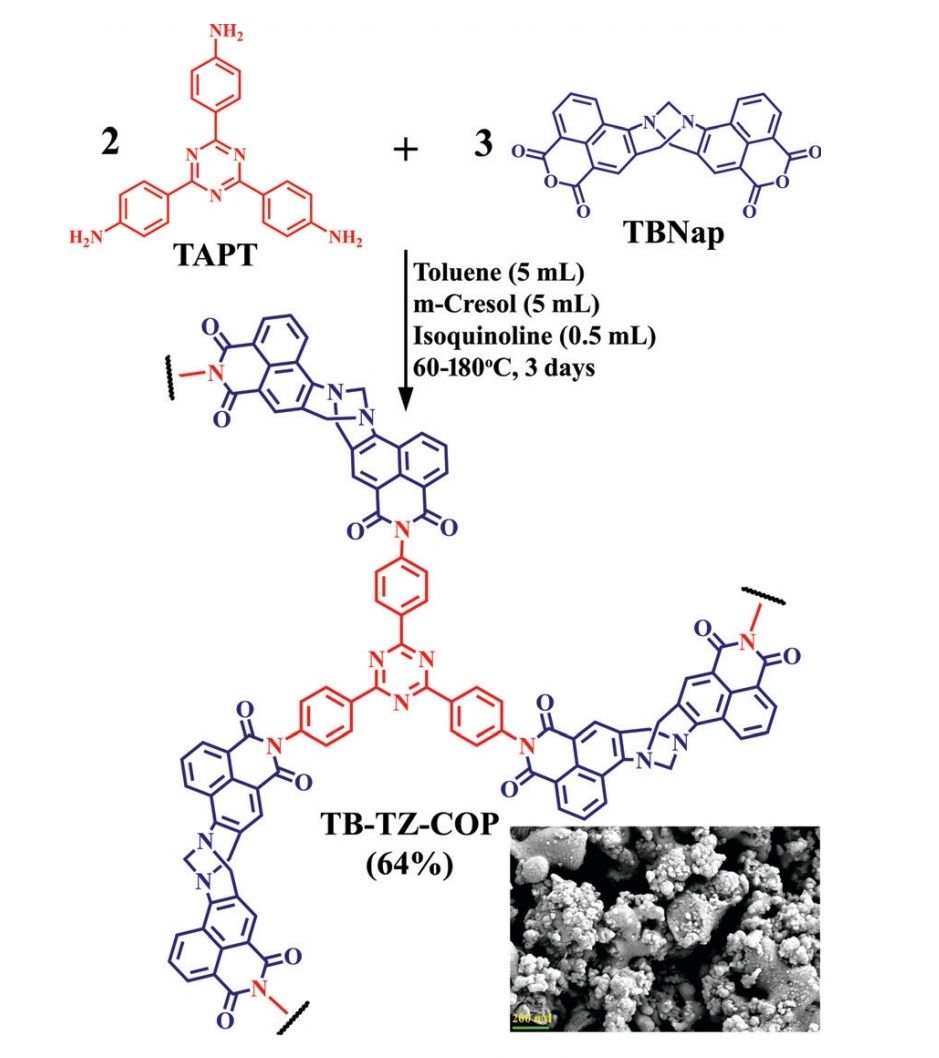
The team set out to find the reason why TB-TZ-COP was able to display different emission intensities, even with closely related VOCs. Initially, it was assumed that the varying amounts of energy transferred from different VOCs to the sensor was responsible for these results. However, this did not seem to be completely consistent with the data collected.
Intermolecular interactions were also known to significantly affect emission intensities (enhancements). Additionally, dioxane was known to have significant interactions with solutes, particularly those with aromatic rings. Armoured with these facts, the team sought to investigate why TB-TZ-COP displayed a high degree of emission intensity for dioxane in particular.
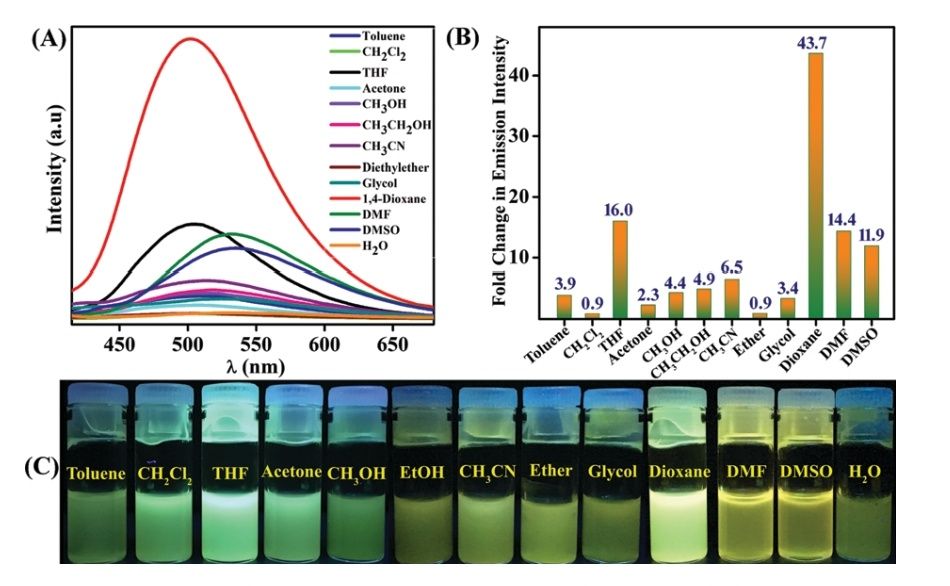
Dioxane, apart from being an environmental pollutant, also happens to be an industrially important chemical. It is generally synthesised through condensation of ethylene glycol. This is where TB-TZ-COP may prove to be particularly useful (i.e. in determining how far the condensation reaction has proceeded, and how much 1,4-dioxane was obtained). The team demonstrated that the emission intensity of TB-TZ-COP in a mixture of glycol and dioxane increased with an increase in the volume percentage of dioxane. Additionally, as the polarity of the solvent decreased (due to the increasing concentration of 1,4-dioxane, a nonpolar molecule), a decrease in wavelength was observed. The fluorescence was clearly visible to the naked eye.
One property commonly desired in fluorescence sensors is for them to be reusable in different environmental conditions with the same level of performance. To verify that TB-TZ-COP satisfied this requirement, the team first recorded its emission intensities in glycol and dioxane. They then isolated the polymer using centrifugation, and measured its emission intensities in the same solvents again. Even after repeated cycles, TB-TZ-COP continued to reproduce the same results, thereby highlighting its reversibility.
Another thing that needs to be taken into account is that VOCs are generally present in the environment in the vapour phase. Hence, testing the capabilities of a fluorescence sensor in the presence of vapours of VOCs is crucial. It was found that a thin film of TB-TZ-COP showed noticeable changes in emission intensity (visible to the naked eye) on exposure to saturated vapours of dioxane.
CONCLUSION:
These results show that the TB-TZ-COP polymer has a lot of potential as a fluorescence sensor, being able to distinguish between glycol and 1,4-dioxane even though they are structurally and chemically similar. TB-TZ-COP was shown to be capable of distinguishing between VOCs in both their liquid and vapour phases. Further research is being done to understand the various properties of such sensors.