How a team from IIT Roorkee was successful in developing the world’s first 'specific bacterial biosensor' for detection of environmental detergent pollutant — SDS (Sodium Dodecyl Sulphate)
Biosensors are novel engineered machines used to measure biomarkers analytically. These markers are a combination of biological receptor compounds (antibody, enzyme, nucleic acid, etc.) that are directed during specific biological events (e.g., antibody-antigen interaction). They allow the detection of a broad spectrum of analytes in complex sample matrices. They have also shown great promise in areas such as clinical diagnostics, food analysis, bioprocess, and environmental monitoring.
SDS (Sodium Dodecyl Sulphate) is one such synthetic organic compound that generates biomarkers. It is extensively used in soaps, toothpaste, creams, shampoos, laundry detergents in households, agricultural operations, laboratories, and industries. But it's notoriously famous for its toxic effects when disposed of in waterways and deteriorating drinking water quality. Therefore it becomes a top priority to create a biosensor that could precisely detect SDS in different samples.

Prof. Naveen K. Navani, Department of Biotechnology, IIT Roorkee, and Sourik Dey, a postgraduate student at the Department of Biotechnology MSc program at IIT Roorkee, acknowledged the threat associated with SDS and decided to work on creating the world’s first-ever state-of-the-art biosensor to measure it. Along with their group-members, Shahnawaz Ahmad Baba, Ankita Bhatt, and Rajat Dhyani, the team successfully created this novel biosensor. But their journey wasn’t easy from the start. Before we dive into their research work, let's understand how to measure a biomarker and the problems associated with it.
Biomarkers are infamous for their extreme tolerance to being tracked by sensory devices. Let me give you a taste of today’s biosensor technology. When an average person sweats, it generates approximately 800 unique biomarkers that can give us feedback on things like muscular exertion, fatigue, hydration level, or electrolytic levels. Yet our modern-day smartwatch bio-sensors can only do one of two things — They can either measure steps or heart rate.
These devices are literally stuck in the Stone Age because they don't have any chemical sensing capabilities.
The modern advent of biosensor technology works on the principle that surfaces are selective to biomarkers. And through the innovations in Electrochemistry, we can translate these markers into either voltages or currents that can be read out on a scoring device. This device could be later imprinted on a mass scale for detecting biomarkers in a wide variety of applications.

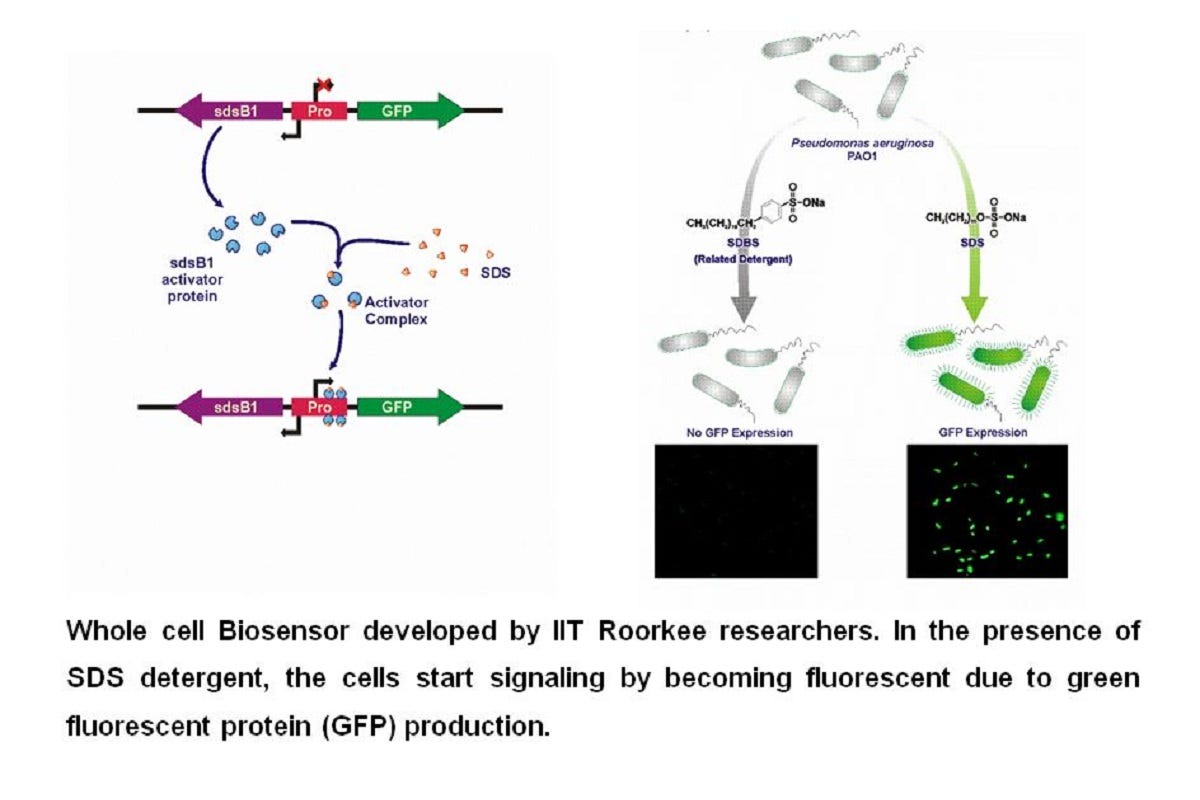
Bio-engineers have been working continuously to find techniques to detect SDS. Unlike conventional methods, SDS can easily be confused with closely related detergents like SDBS (Sodium Dodecylbenzenesulfonate). The IIT Roorkee team came with the idea of using Pseudomonas Aeruginosa PAO1 strain as the framework (chassis) to construct a whole-cell biosensor for detection. The system involves a highly specific regulator and a fluorescent protein produced only when SDS is present in the sample.
The system can even detect 0.1 ppm of SDS in aquatic samples!
This biosensor is highly specific for SDS and has minimal interference from other detergents, metals, and inorganic ions present in the environment. To get a deeper insight into this unique approach, we asked a series of questions with the team.
1. How does the Biosensor work and accurately able to predict SDS?
Pseudomonads have an inherent capability to be used as an optimal destination framework for synthetic biology applications. The selected species of Pseudomonas can be engineered to detect various chemicals owing to their resilient nature to survive and adapt to harsh environmental conditions. This research highlights the development of the world’s first whole-cell bacterial biosensor for the direct, specific, and efficient detection of SDS without involving sample preparation steps, toxic chemicals, sophisticated polymers, and sensor development steps.— Sourik Dey, Final year MSc student at IIT Roorkee.
2. What makes your biosensor different from other conventional approaches?
SDS, if untreated, can harm the marine biodiversity and cause pollution of land and water-bodies. The highlight of this biosensor is its sensitivity to even minute quantities of SDS in the environment and its ability to distinguish between SDS and SDBS. — Prof Naveen Kumar Navani, Department of Biotechnology, IIT Roorkee.
3. What makes SDS so dangerous?
The constant deterioration of the quality of drinking water and the harm caused to marine life are some of the major concerns. SDS has harmful effects on the survival and breeding of organisms in the aquatic ecosystem. It hampers their biological processes, such as solubilization of phosphate, reduction of ammonia, nitrogen fixation, and photosynthesis. It can cause dermal and ocular irritation, cardiac anomaly, hemolysis, tachycardia, kidney failure, and even death. SDS can also disrupt biological wastewater treatment processes and cause problems in sewage aeration and treatment facilities owing to its high foaming capacity. — Prof Naveen Kumar Navani

4. What are the applications of your biosensor?
Due to the specificity of this Biosensor, it can show accurate and precise analysis of sewage water plant treatment. This biosensor's genetic modification to produce green fluorescence signals gives it an edge over other conventional biosensors. It can be easily installed at various inlet and outlet pipelines of sewage plants to determine SDS concentration. Moreover, the biosensor can be easily grown and distributed so that people can test SDS concentration in water samples from different samples. — Sourik Dey
5. What role can biosensors play in the future of diagnostics?
Bio-sensors have the capability to detect various kinds of chemical receptivity and give real-time data analysis. Early-stage lung cancer, one of the most common and aggressive cancers, kills around 1.4 million people worldwide every year, so the pursuit of new techniques to detect it accurately remains a global challenge. Now, a highly sensitive graphene biosensor has shown potential in electronic nose devices, which analyze the components of vapor mixtures such as the breath. Artificial intelligence could soon be used to predict the spread of melanoma by using microscopic cameras to analyze the appearance and behavior of cells. Robotic sensor technology that can be used to measure hormones quickly and cheaply could pave the way for the diagnosis of reproductive health issues in real-time. The future holds various possibilities for the application of biosensors. — Prof Naveen Kumar Navani
Conclusion
Biosensors represent the technological counterpart of living senses. These technologies have already reached a highly advanced level and need minor improvement at most. They have the potential to substitute antibodies in biomarkers and biochips for the measurement of low-molecular-weight substances, proteins, viruses, and living cells.
The dream of the “100-dollar” personal genome may come true in the next few years, provided that the technological hurdles of nanopore technology or of polymerase-based single-molecule sequencing can be overcome.
In the future, individual therapy will include genetic profiling of isoenzymes and polymorphic forms of drug-metabolizing enzymes. For decentralized online patient control or the integration into every day “consumables” such as drinking water, foods, hygienic articles, clothing, or for control of air conditioners in buildings and cars and swimming pools, a new generation of “autonomous” biosensors will emerge.



