Team iGem IIT Roorkee’s gold medal solution to tackling WHO’s topmost priority problem of Antimicrobial Resistance and approaching it from a multidisciplinary perspective.

The World Health Organization describes Antimicrobial Resistance (AMR) as the most critical and urgent problem to address today. At the 68th World Health Assembly in May 2015, a global action plan was endorsed to tackle antimicrobial resistance, including antibiotic resistance, the most urgent drug resistance trend. Still, today’s statistics show that about 35–40% of all hospital admissions in India contract hospital-acquired infections due to Multidrug Resistance with no pertinent treatment. The corollary is that the hospital environment is one of the biggest reservoirs of antibiotic-resistant pathogens.
Team iGEM IIT Roorkee believes that in the coming decades when bacterial infections would become untamed due to drug resistance, we are likely to witness a ten-fold increase in death rates and indefinite damage to the global economy in comparison to the pandemonium caused by the current coronavirus pandemic. The situation becomes more acute due to the naïve outlook of people towards the intake of antibiotics. The team felt an imperative need to build alternative solutions to treat bacterial infections and reduce healthcare systems’ dependence on antibiotics. With the theme ‘How to break the Antibiotic Dependence of the healthcare systems and control the damage caused?’, the team presented their solution at the iGEM competition and also won the Gold medal for their exceptional work.

The iGEM Foundation (International Genetically Engineered Machine) is an independent, non-profit organization dedicated to the advancement of synthetic biology, education and competition, and the development of an open community and collaboration. The iGEM Competition gives students the opportunity to push the boundaries of synthetic biology by tackling everyday issues facing the world.
I aim to illustrate the technical and intricate workings of their project to you and provide insight into how to perceive antibiotic awareness in our daily lives. But before moving forward, let’s ask the question, “ What causes antibiotic resistance?” Antibiotics are nothing new to us. We take them for all purposes. From minor infections to serious health issues caused by bacteria, we depend upon them. They essentially neutralize the bacteria by interrupting cell wall synthesis or interfering with vital processes like protein synthesis. And it does it all while keeping our cells unharmed. But as our dependencies start increasing on them, the bacteria start to become immune to the treatment.
Darwin’s theory of natural selection fundamentally tells us that individual bacteria can undergo random mutations giving them an edge in survival. They learn from our antibiotics and engineer an immunity profile in their DNA, and help other bacteria fight against the antibiotics. As we intake more and more antibiotics for our small needs, the bacteria become more resilient towards it and eventually gain resistance to any future treatments. To give you an overview of the resistance levels attained by the present scenario, bacteria, Staphylococcus aureus, the primary bacteria causing Pneumonia, has reached its mutation stage and is immune to most treatments. This makes the treatment of an infected person compound and complex. So how do we go about attacking a problem that involves the breakdown of the DNA structure in these bacteria?
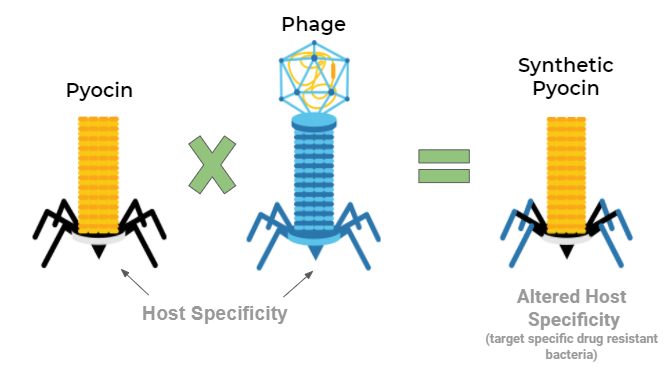
The research division at iGEM IIT Roorkee studied the problem extensively and found that the solution lies in a narrow-spectrum treatment for the multidrug-resistant pathogen, Acinetobacter baumannii, by the engineering and combination of natural antibacterial molecules-bacteriophages and pyocins. This results in the creation of a novel antimicrobial protein complex called Seekercins. The team essentially created a bioengineered fusion of naturally occurring antibacterial molecules and synthetic antibiotics to create an advanced host to attack bacteria. The task requires intensive bioinformatics study upon different biomolecules and their behavior to various stimuli. The team used a machine learning approach called DARG, i.e., Detection of Antibiotic-Resistant Genes, to provide faster and more accurate detection of the compatible antibiotic-resistant genes.
Team iGEM IIT Roorkee tells us that they performed multiple sequence alignment of resulting bacteriophage tail fiber sequences. The task required choosing the right bacteriophage and the correct plasmid design to hold them.

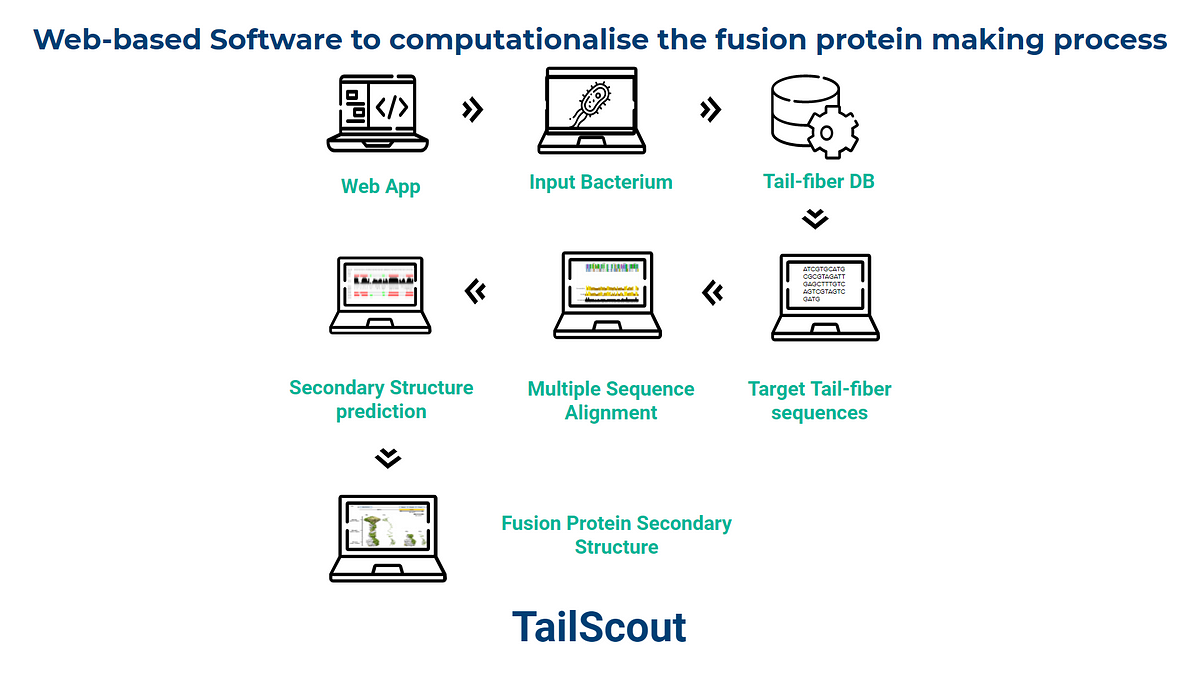
The team also designed a web-based software tool, called TailScout, for conveniently designing these fusion proteins. The aim was to execute the computational process and the rendering of the Seekercin on a better-controlled environment. After successfully identifying the resistant class of genes, the team encapsulated their behavior into a single unit for integrated human trials.
The team tells us that they gained several insights on AMR infections during COVID-19 across India and also understood the patients’ mental and physical trauma to give purpose to the project.
They believe that their approach is capable of giving humanity a step ahead of these notorious bacteria. The novel working relationship with natural antibacterial resistors is a cutting edge idea alongside phage therapy and vaccines. But the team also feels that the real solution lies in the habits we follow. And to give their project a significant meaning, they collaborated with National Service Scheme, IIT Roorkee, to host the antimicrobial awareness week at IIT Roorkee. They organized several webinars, quizzes, and published infographic posts to inform people of the threats we face and what we can do to fight antimicrobial resistance.
The Team Leader, Sanjeevani Marcha, says: “We believe that through our concept and protein design, we can focus on the priority pathogens and urge the pharmaceutical companies to change the ways antibiotics are conventionally developed. By working together on a biological system with an engineering approach, we could think beyond traditional methods in life sciences and implement creative approaches from an open mindset.”
iGEM IIT Roorkee won the Gold medal at the iGEM competition held in November 2020. Through their project Pyomancer, they propagated the message that the time has come for a paradigm shift to narrow-spectrum antimicrobial therapies in the healthcare sector to regulate the damage done by the continuous use of broad-spectrum antibiotics. This new class of Antimicrobial Complexes is our valuable arsenal at fighting the battle against pathogenic bacteria. Scientists are still pursuing better ways of understanding the abstract structure of these bacteria. And we hope for better treatments for bacterial infections in the future. But the immediate long-term treatment lies with us! It lies in our perception of the antibiotics we consume and the awareness we have about them.



