Emmanuelle Charpentier of France and Jennifer Doudna of USA, on October 7, won the Nobel Prize in Chemistry “for the development of a method for genome editing” technique known as the CRISPR-Cas9 DNA snipping “scissors” that can cut DNA at a precise location, allowing scientists to make specific changes to specific genes. This year’s prize is special as it is the first time a Nobel science prize has gone to a women-only team. They have become only the 6th and 7th women in Chemistry to win a Nobel Prize.

About the Nobel prize winners:
Emmanuelle Charpentier is a French professor and researcher in microbiology, genetics, and biochemistry. Since 2015, she has been a Director at the Max Planck Institute for Infection Biology in Berlin.
Jennifer Anne Doudna is an American biochemist with a vast knowledge of RNA. She is a Chair Professor in the Department of Chemistry and the Department of Molecular and Cell Biology at the University of California, Berkeley. She has been an investigator with the Howard Hughes Medical Institute since 1997.
How they found their discovery:
During Emmanuelle Charpentier’s studies of Streptococcus pyogenes, one of the bacteria that cause the most harm to humanity, she discovered a previously unknown molecule, tracrRNA. Her work showed that tracrRNA is part of bacteria’s ancient immune system, CRISPR/Cas, that disarms viruses by cleaving their DNA.
Charpentier published her discovery in 2011. The same year, she initiated a collaboration with Jennifer Doudna. Together, they succeeded in recreating the bacteria’s genetic scissors in a test tube and simplifying the scissors’ molecular components, so they were easier to use.
When bacteria survive a viral attack, they incorporate snippets of the virus’ DNA into their genetic material or genome. Those stolen segments, and the genetic padding between them, are called Clustered Regularly Interspaced Short Palindromic Repeats, or CRISPS.
If the virus attacks again, the bacteria use those CRISPR segments as a template to create strands of RNA that fixate on a strand of resembling sequence in the virus’ genome. The CRISPR RNA carries along a protein called Cas9 to the target location of the DNA. Cas9 disarms the virus by cutting its DNA at the target location.
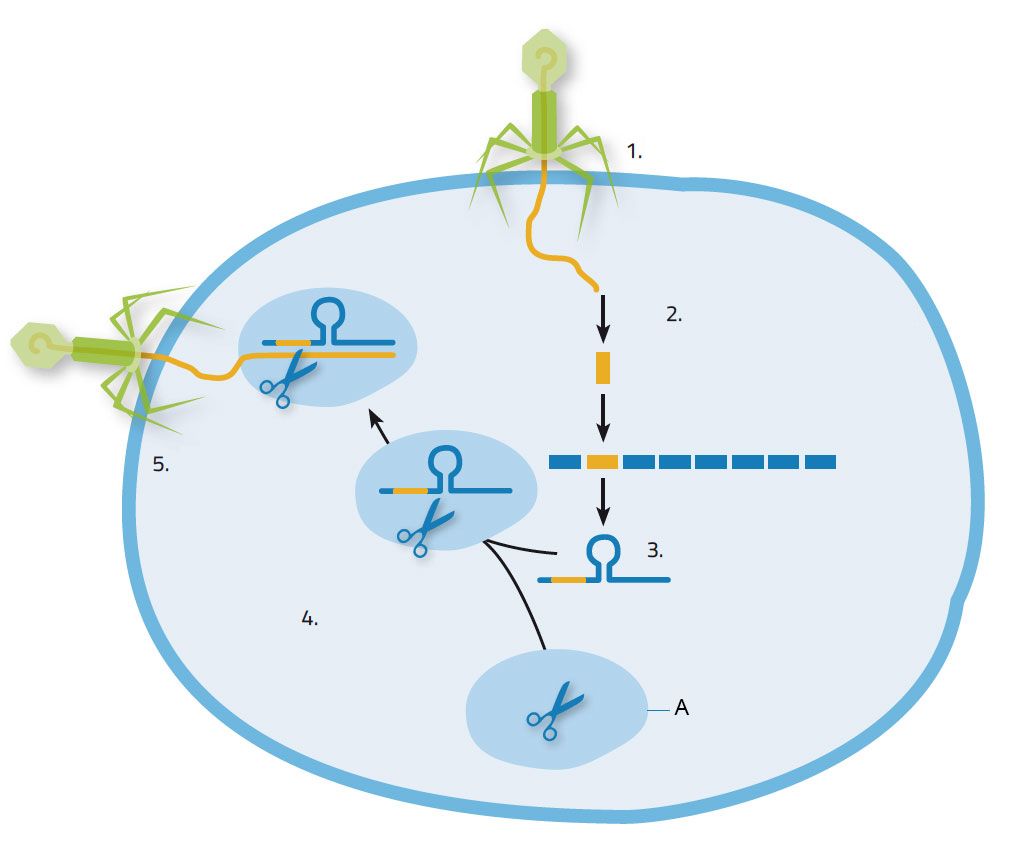
Emmanuelle Charpentier and Jennifer Doudna were amazed by this clever bacterial immune system and wondered if they can reprogram it to make cuts, or edits, to any DNA. The two scientists created a new molecule called guide RNA, which can easily be customized to mirror any DNA strand.
The custom guide RNA (tracrRNA) molecule is like a tracker. It steers Cas9, the molecular scissors, to cut a precise portion of the DNA. With CRISPR-Cas9, a researcher can now see what happens when they turn off a gene by cutting it. They can even program the CRISPR-Cas9 complex to insert a new DNA sequence to repair or edit a gene. The technique has become widespread in labs since Charpentier and Doudna first described it in 2012.
Impact of their discovery:
Since Charpentier and Doudna discovered the CRISPR/Cas9 genetic scissors in 2012, their use has exploded. This tool has contributed to several important discoveries in basic research, and plant researchers are ready to develop crops that withstand mould, pests, and drought. In medicine, clinical trials of the latest cancer therapies are underway, and therefore the dream of having the ability to cure inherited diseases is close to coming true. These genetic scissors have taken the life sciences into a replacement epoch and, in some ways, are bringing the best benefit to humankind.

