Did you know that the formation of a cancerous tumor can be seen outside (in-vitro) the human body? One of the authors of the research paper “Transport of vascular endothelial growth factor dictates on-chip angiogenesis in tumor microenvironment”, Jyotsana Priyadarshini, talks about how a cancer tumor and its working can be observed and controlled on a “chip”.
Jyotsana is currently pursuing research in the area of organ-on-a-chip, and addresses the important features and geometrical complexities of microvasculature, i.e., how blood vessels and flow function, including their structural features, and also how disease can spread from host site to different sites in the body (metastasis). She has also valuably contributed to other research papers in this domain. She explains that such bioengineered in-vitro microfluidic platforms are potentially suitable for probing the way cells interact and offer critical insights to cancer, cardiovascular
diseases and drug discovery. Instead of testing on animals or on petri dishes, the chip can be designed to have striking resemblance with the human physiological system.
It can be done using a system called “organ-on-a-chip” (OOC). As the name suggests, these chips can mimic the key features of different organs and human physiological conditions, by culturing human-derived tissues inside a bioengineered “microfluidic” chip.
Microfluidics deals with small amounts of fluid that can be precisely controlled in smaller channels having diameter range between 10-100 μm. The OOC of concern here is related to a diseased condition called “tumor angiogenesis” and surrounding environment altogether termed as “tumor microenvironment”.
A tumor microenvironment is the environment surrounding the tumor, that include blood vessels, immune cells, fibroblasts, signaling molecules and the extracellular matrix. The functional microfluidic chips may help imitate the microenvironments of the tumor and maintain the tissue-specific functions by controlling the fluidic conditions.
Further elaborating on the term extracellular matrix (ECM) which is mentioned above, it is a network of proteins and other molecules that surround, support and give structure to cells and tissues in the body. The ECM helps cells attach to, and communicate with, nearby cells, and plays an important role in cell growth, cell movement, and other cell functions.
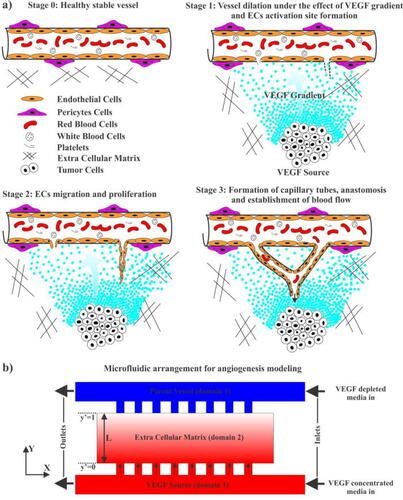
Tumor cells consume a lot more oxygen and nutrients, compared to a healthy cell, leading to a “hypoxic state”. A hypoxic state is where a cell doesn’t receive sufficient oxygen to carry out its regular functions. To overcome this state, the tumor cells produce a few chemicals, which diffuse in ECM space creating a chemical gradient. Out of tumor-produced chemical agents, vascular endothelial growth factor (VEGF) plays a major role for enhancing the generation of new blood vessels branched from a nearby parent vessel to supply the oxygen and nutrients to the tumor source. The new capillaries are mainly formed using endothelial cells (ECs), by migration and spread of the same.
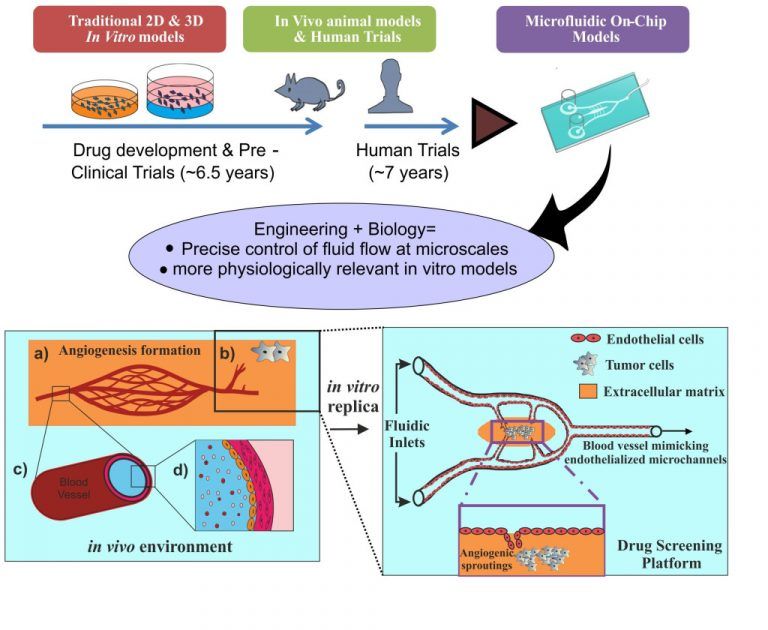
The concentration differences in VEGF stimulates that nearby blood vessel to sprout new capillaries (a process called angiogenesis), buildup of ECs toward higher concentration of VEGF, i.e., towards the tumor. The VEGF is produced by tumor cells and the chemical gradient help movement of capillaries up to reach the tumor cell. Other than this well-established theory, what was not accounted for was the role of leakage of fluids, called interstitial flow, how it affects the establishment of chemical gradient and chemical migration is one of the issues that is addressed in this research paper. This leakage creates a complicated surrounding, and would make it even more difficult to target the tumor cell for drug delivery. This comprehensive modeling integrated the computational framework of fluid transport and continuum mathematical model of angiogenesis with designing of OOC to help study and control the process of angiogenesis more closely since this is the basis of growth of a tumor.
Jyotsana, along with one of her fellow research authors, designed the computational model at home, when there were COVID-19 restrictions.
What was her motivation to take up this subject of interest? She says,
“...animal free research! Basically, the conventional methods, you could say, disease modeling and drug testing, take place in small animals. The animals’ genotype and physiology completely vary from human one. When a drug comes into the market, it goes through pre-clinical trials, like 2d-3d petri dish(failed-static). Very long-5 to 6 years, generally 9 out of 10 will fail. Then goes for several human trials.”
She wanted to bridge this gap with microfluidic chips. She continues to say, “And organ complexity is there, and specificity will be lacking there. So, in a microfluidic chip, you can provide the dynamic and control the fluid flow.”
The design developed by Jyotsana and team gives us a deeper insight into how a tumor microenvironment functions, and what one should also take into account to deliver the drug more efficiently to the tumor cell, taking the medical community one step closer towards curing cancer once and for all!

